X-RAY RUNS: Apply for Beamtime
2017 Nov 1 - Dec 21
2018 Feb 7 - Apr 3
2018 Proposal/BTR deadline: 12/1/17
2018 Apr 11 - Jun 4
2018 Proposal/BTR deadline: 2/1/18
Serge Desgreniers and Ken Lagarec, Laboratoire de
physique des solides densesUniversity of Ottawa, Ottawa-Carleton
Institute of Physics, Ottawa, Canada [Phys. Rev. B, Vol 59, 8467(1999)]
Zirconium dioxide and hafnium dioxides quenched at room conditions from
high pressure have yielded ultra dense, low compressibility crystalline
forms of matter. These materials are of interest for their potentially
large hardness (resistance to deformation). The x-ray measurements below
complement the optical, electrical, and transport properties measured in
the home laboratory to give a more complete picture about the properties
that describe these unusual materials.
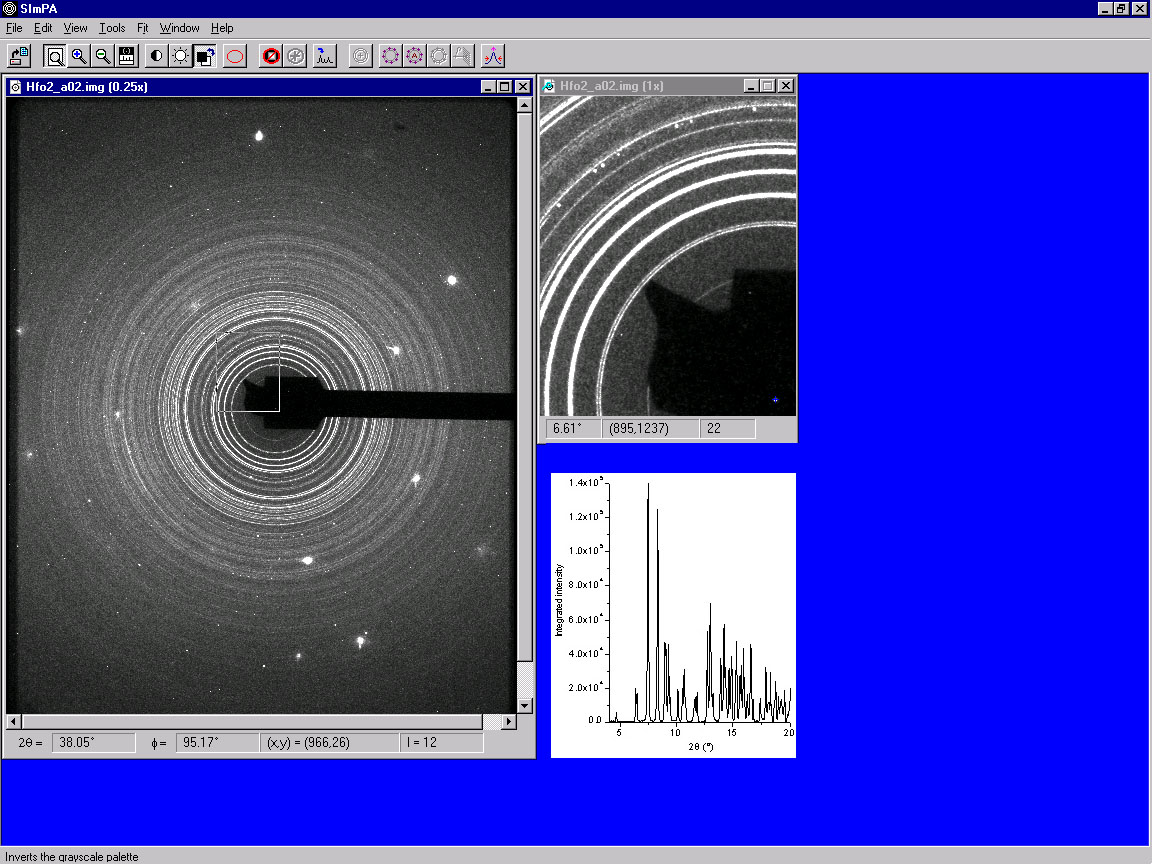
Angle dispersive x-ray diffraction is a key technique to study the structural
modifications in dense materials. At the left are the Debye rings from the x-ray
diffraction pattern of hafnium oxide at a pressure of 1.2 GPa in a diamond anvil cell,
taken at the CHESS B2 station. The spacing of the rings depends on the actual distance
between atoms, a value that changes in response to the pressure generated in the diamond
anvil cell.
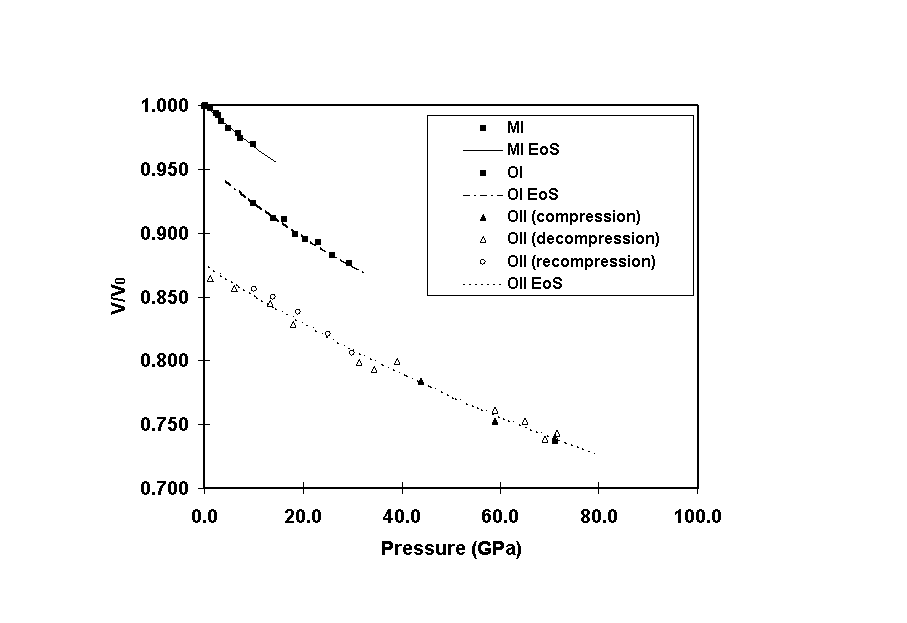
The relative volume change with pressure (equation of state) has been obtained from the Debye ring pattern above for the dense phases of hafnium oxide. The high density phase, retrieved at room conditions from a pressure exceeding 70 GPa, presents a relatively high bulk modulus (B0=340 GPa) with a relative volume reduced to 0.87.
Other studies of dense oxides, as potentially ultrahard materials are underway. The ability to perform diffraction measurements at CHESS is a key aspect to the success of the understanding of strong atomic bonding in dense oxides and how it translates into hardness.