X-RAY RUNS: Apply for Beamtime
2017 Nov 1 - Dec 21
2018 Feb 7 - Apr 3
2018 Proposal/BTR deadline: 12/1/17
2018 Apr 11 - Jun 4
2018 Proposal/BTR deadline: 2/1/18
G.R. Hoffman, N. Nassar, and R.A. Cerione
Departments of Molecular Medicine, and Chemistry and Chemical Biology,
Vet Medical Center, Cornell University, Ithaca, New York
[Cell, Vol 100, 345-356 (2000).]
Cancer researchers have made a structural map of cellular switches
called Cdc42 complexed with GDI(guanine nucleotide-dissociation
inhibitor). The structural mapping was obtained with x-ray
crystallographic methods from data collected with MacCHESS resources at
CHESS. MacCHESS, a NIH supported resource, builds and operates the
specialized x-ray detectors and cameras needed to collect the data from
crystals grown of this complex.
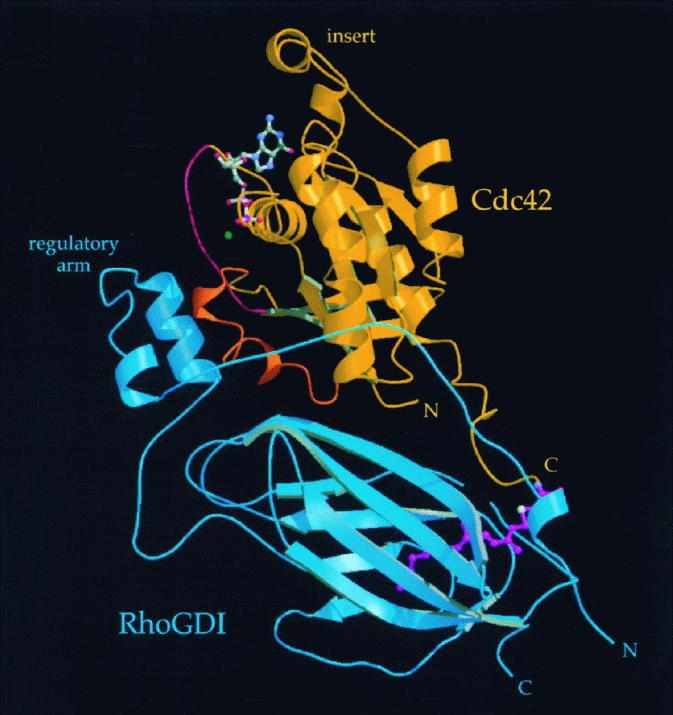
The results of the diffraction analysis are given in the figure at the
right. Here groups of atoms are shown as ribbons of connected structure.
The regulatory arm (blue) of the structure binds to switch I (pink) and
switch II (orange) regions, giving the researcher a three-dimensional
physical picture of the inhibited state.
Additional biochemistry studies [1] show how a hyperactive form of the
Cdc42 molecular switch can increase the shuttling of other proteins
throughout the cell thus disrupting the orderly cell growth. If the
Cdc42 part is mutated and switched on for an extended period of time,
the cellar activities are overstimulated, thus producing malignant
cells, i.e. cancer.
This discovery offers potential targets for drugs to block tumor growth.
This research was made possible by the operation of CESR (Cornell
Electron – Positron Storage Ring) a NSF supported high-energy storage
ring designed for particle physics and CHESS, a NSF laboratory
supporting both material science and biological work. This work
illustrates how important integrated facilities with various
interdisciplinary staff are contributing to ongoing fundamental cancer
research.
1. W.J. Wu, J. W. Erickson, R. Lin, & R. A. Cerione, Nature, Vol 405,
15 June 2000, 800-801.
