X-RAY RUNS: Apply for Beamtime
2017 Nov 1 - Dec 21
2018 Feb 7 - Apr 3
2018 Proposal/BTR deadline: 12/1/17
2018 Apr 11 - Jun 4
2018 Proposal/BTR deadline: 2/1/18
The vacuolar ATPase (V-ATPase) is a large, multi-protein enzyme complex that pumps acid (protons) across lipid membranes in all animal and plant cells. It uses the main energy currency of the cell (adenosine triphosphate (ATP) via hydrolysis) to transport protons across biological membranes, a process that results in acidification of intracellular compartments (organelles) or the extracellular space. The acidic environment provided by the V-ATPase is essential to a variety of both normal and disease processes including pH homeostasis, urinary acidification, insulin secretion and tumor metastasis. In line with its contribution to fundamental cellular processes, the V-ATPase is highly conserved from yeast to humans.
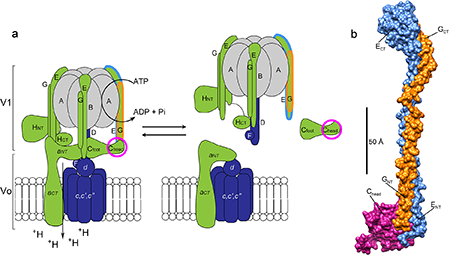
Figure 1: V-ATPase regulation and the X-ray structure of the EGC(head) complex. (a) Schematic of V-ATPase regulation by reversible dissociation showing the active assembled enzyme (left) and inactive dissociated enzyme (right). The rotary subunits are shown in purple, static subunits in green and the catalytic hexamer in gray. The static subunits prevent rotation of the hexamer during catalysis. The peripheral stalk EG heterodimer and regulatory C subunit (head domain) are outlined in blue, orange and pink according to their coloring in the 2.9 Å resolution crystal structure shown in (b). The structure (b) of the EGC(head) complex is highly elongated (150 Å) with the regulatory C subunit bound on one end of the parallel EG heterodimer.
The activity of the complex is regulated by a unique mechanism in which the part of the complex responsible for ATP hydrolysis (V1) detaches from the membrane embedded proton channel (Vo) in a process referred to as reversible dissociation (Figure 1). The V-ATPase is a biological rotary motor: conformational changes induced by ATP hydrolysis taking place on the catalytic hexamer (A and B subunits) are coupled to the proton channel by a rotating central stalk, or axle, of the motor. The static part of the motor includes three elongated peripheral stalk heterodimers (subunits E and G) that mediate a number of important contacts with other protein subunits [1]. Specifically, the interactions involving the peripheral stalks must be broken and re-formed during the unique mode of enzyme regulation known as “reversible dissociation”. Indeed, when the activity of the V-ATPase is not needed, the enzyme undergoes a dramatic (but fully reversible) structural rearrangement wherein the soluble (V1) and membrane (Vo) subcomplexes disengage from one another. Interestingly, while dissociated, a single enzyme subunit (subunit C) is released from both subcomplexes and reincorporated during re-assembly [2].
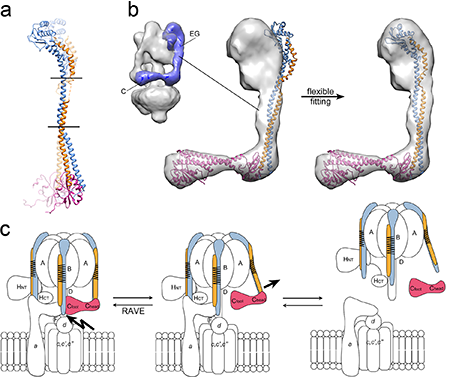
Figure 2: Flexibility and spring loading of the peripheral stalks. (a) Alignment of the structures of the two conformations of the EGC(head) complex (E blue, G orange and C(head) pink) with horizontal lines showing the position of the two flexible hinge regions in the EG coiled-coil. (b) On the left is an electron microscopic (EM) reconstruction of the V-ATPase with the density belonging to the EGC complex in purple. Fitting of either conformation of the EGC(head) complex (aligned with the structure of the intact C subunit) into the segmented EM map revealed a mismatch (middle panel). After flexible fitting (right panel), the complex fit nicely into the map by bending along the hinge regions identified by comparison of the two conformations shown in (a). (c) Schematic of the initiation of enzyme regulation by reversible dissociation. (Left) A chaperone complex (RAVE) inserts subunit C into the V1-Vo interface for enzyme assembly, thereby inducing tension into and spring loading the EG peripheral stalks. (Middle) Upon the signal(s) for enzyme dissociation an interaction with subunit C is modulated thereby releasing the tension in the peripheral stalks and initiating the disassembly process. (Right) The inactive, fully disassembled complex.
What is the structural basis for the reversible dissociation - in other words, how is the detachment of the V1 from the Vo initiated? A key piece of the puzzle was recently provided by the X-ray crystal structure of the peripheral stalk subunits (EG) connected to the regulatory C subunit (head domain) (Figure 2). The study was performed by Rebecca Oot in the Wilkens and Berry labs at SUNY Upstate Medical University using data collected at CHESS beamlines A1 and F1, and the results are reported in the journal, Structure [3]. Two crystal structures of this complex were solved, revealing two different conformations of the proteins. These structures represent the first high resolution (2.8 and 2.9 Å) structures of a sub-complex from the eukaryotic V-ATPase and importantly involve an interaction that must be broken during enzyme regulation. The structures show that the peripheral stalk subunits (EG) are highly elongated (100 Å) and flexible, with the C subunit bound on one end. A comparison of the two conformations revealed two flexible hinge regions flanking the middle portion of the EG coiled-coil. A fit of the crystal structures into an electron microscopic reconstruction of the intact enzyme revealed a mismatch, indicating that the EG-C (head) complex is in a more open conformation than would be required when assembled into the intact enzyme. Using a flexible fitting algorithm to assess the inherent flexibility of the protein complex and the ability of the crystal structures to fit into the intact enzyme indicated that bending along the hinge regions would allow for accommodation of the sub-complex into the intact enzyme. As a chaperone known as the RAVE complex is required for reassembly of the enzyme during regulation, the Wilkens and Berry groups propose that this chaperone positions the C subunit into the V1-Vo interface thereby spring-loading the peripheral stalk and preparing the motor for rapid and efficient dissociation when needed.
References:
[1] Oot, R. A and Wilkens, S (2012). Subunit interactions at the V1-Vo interface in yeast vacuolar ATPase. J Biol Chem 287, 13396-406.
[2] Oot, R. A and Wilkens, S. (2010) Domain characterization and interaction of the yeast vacuolar ATPase subunit C with the peripheral stator stalk subunits E and G. J Biol Chem 285, 24654-64.
[3] Oot, R. A., Huang, L. S., Berry, E. A and Wilkens, S. (2012) Crystal structure of the yeast vacuolar ATPase heterotrimeric EGC(head) peripheral stalk complex. Structure 20, 1881-92.
Submitted by: Chae Un Kim, MacCHESS, Cornell University
4/16/2013
