X-RAY RUNS: Apply for Beamtime
2017 Nov 1 - Dec 21
2018 Feb 7 - Apr 3
2018 Proposal/BTR deadline: 12/1/17
2018 Apr 11 - Jun 4
2018 Proposal/BTR deadline: 2/1/18
Water has an unusual property at cryogenic temperatures; it becomes randomly arranged in a way similar to glass. This property, known as the amorphous state, is important in X-ray crystallography because protein crystals need to be maintained at low temperatures without having its lattice disrupted by crystalline ice. Conventional methods use liquid nitrogen to cool the protein crystals (with cryoprotectants) so that low-density amorphous (LDA) ice, rather than crystalline ice, forms. Kim et al. developed a procedure known as high-pressure cryo-cooling that uses high-pressure on protein crystals to form high-density amorphous (HDA) ice instead [1].
Both HDA and LDA are desirable for x-ray crystallography since water molecules are randomly arranged in either state. However, little is known about how water molecules transit between HDA and LDA states. A recent study by Chae Un Kim (a former MacCHESS staff scientist), Mark W. Tate (Senior Researcher Associate of Gruner’s group) and Sol M. Gruner (Professor in Department of Physics) induced HDA by cryocooling dilute aqueous solutions at high pressure of 2000 bar. Then they introduced cracks in the solution samples by applying mechanical stress on the sample. When the HDA samples were warmed from 80 K at 1 bar, they observed that cracks began to heal at 120 K, suggesting the HDA-to-HDL (high-density liquid) transition (Fig 1). They further investigated the phase behavior of the high-density states using time-resolved X-ray wide angle scattering (performed at A1 and F1 stations), and found that the high-density states completely convert to a low-density state at thermodynamic equilibrium limit. They also showed that the high-density to low-density phase transition does not include intermediate states other than the two different density states (Fig 2). These observations suggested that the phase transition is a first-order phase transition. The result, which used data collected at CHESS, was published in the issue September 22 at Proceedings of the National Academy of Sciences, USA (PNAS) [2].

Figure 1: Observation of crack healing HDA state upon warming. (Top) Crack healing within the 0.9 M NaK tartrate solution prepared in the HDA state is observed to begin just above 120 K. (Bottom) Crack healing within the 1.5 M NaCl solution prepared in the HDA state is also observed around 120 K.
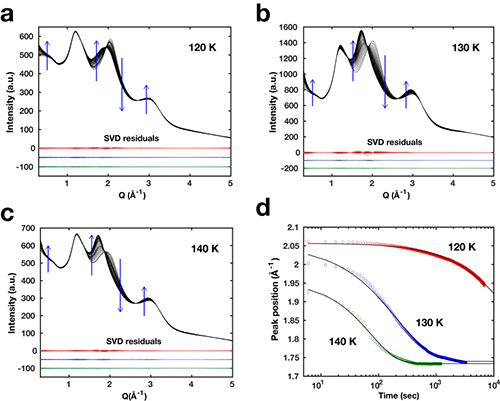
Figure 2: Time-resolved X-ray diffraction study on the phase transformation of 1.5 M NaCl solution from the HDA to the LDA state at three fixed temperatures (120, 130, 140 K). (a-c) Water diffuse diffraction (WDD) profiles during the HDA to LDA transition. Blue arrows indicate the trends for increasing time. Singular value decomposition (SVD) analysis shows that the HDA-LDA transition involves 2 major states. (d) Primary WDD peak positions over time when HDA samples are warmed from 80 K at 6K/min and then equilibrated at 120 K (red), 130 K (blue), and 140 K (green). The data fitted to Avrami-Kolmogorov equations (black solid lines) show that the HDA state progresses to the LDA state (Q=1.7-1.75 Å-1) at fixed temperatures.
Chae Un Kim (now an assistant professor at Ulsan National Institute of Science and Technology (UNIST) in South Korea) said that “These findings support theories that high-density and low-density states are thermodynamically distinct and that they are continuously connected to two different liquid states of water” and “Our results provide insight on establishing delicate theoretical models of water, which will have huge impacts on not only fundamental sciences such as physics and chemistry but also on applied sciences such as mechanical and bio-engineering”.
References:
[1] Chae Un Kim, Raphael Kaper, Sol M. Gruner, High-pressure cooling of protein crystals without cryoprotectants, Acta Crystallogr D Biol Crystallogr. (2005) 61:881-90.
[2] Chae Un Kim, Mark W. Tate, Sol M. Gruner, Glass-to-cryogenic-liquid transitions in aqueous solutions suggested by crack healing, Proc. Natl. Acad. Sci. USA (2015) 112, 11765–11770.
Submitted by:
Teck Khiang Chua, MacCHESS, Cornell University and Chae Un Kim, UNIST, Korea
10/13/2015
