X-RAY RUNS: Apply for Beamtime
2017 Nov 1 - Dec 21
2018 Feb 7 - Apr 3
2018 Proposal/BTR deadline: 12/1/17
2018 Apr 11 - Jun 4
2018 Proposal/BTR deadline: 2/1/18
The CHESS wiggler stations have certainly been among the
most, if not the most productive macromolecular beamlines in existence.
For more than a decade MacCHESS and CHESS have pioneered many aspects of
modern protein crystallography at storage rings and have helped catalyze
the present biostructure revolution. Structural biology at CHESS has
been tremendously successful and now utilizes three out of our four
highest intensity wiggler stations. These stations are oversubscribed by
about a factor of three.
To help satisfy the growing demand for crystallography facilities, the
National Institute of General Medical Sciences (NIGMS) has provided
funds to CHESS to upgrade the high intensity A1, F1 and F2 experimental
stations. The goal of this multiyear upgrade program is to provide
biological user with higher quality x-ray beams and facilities – first
and foremost to deliver x-ray beam with higher intensity, better energy
resolution, tunability and stability. During 1999 we are redesigning and
replacing the x-ray optics room, housings, and monochromator optics on
the F1 and F2 stations. A new collimating mirror will filter the high
power white beam and reduce the enormous heat load on the x-ray
monochromator crystals. Additional developments in x-ray beam
diagnostics and position stabilization with feedback are planned. A
faster computer network and more reliable software and computer
equipment will enhance data collection.
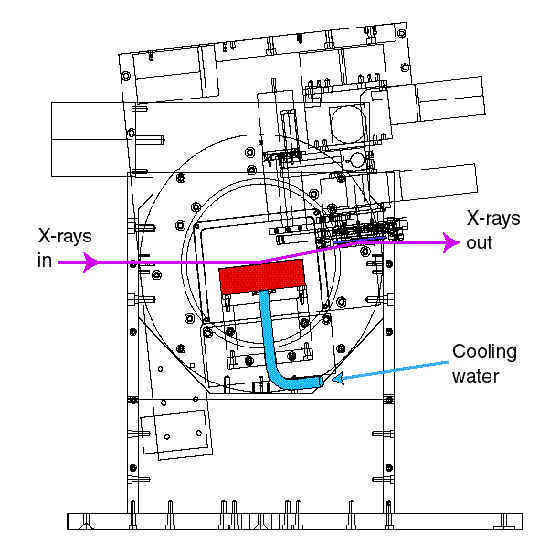
Schematic drawing of the new F2 monochromator
optimized for fast energy tunability and stability needed to improve
Multiwavelength Anomalous Diffraction (MAD) data collection. The
first crystal must be water-cooled to withstand large x-ray heat
loads.
(Drawing: A. Pauling)
