X-RAY RUNS: Apply for Beamtime
2017 Nov 1 - Dec 21
2018 Feb 7 - Apr 3
2018 Proposal/BTR deadline: 12/1/17
2018 Apr 11 - Jun 4
2018 Proposal/BTR deadline: 2/1/18
_______________________________________________

Teacher Peers Through Wavy Glass Capillaries
“Working with glass” is what high school teacher Courtney Couvreur decided to do the first summer after finishing a masters degree program in secondary education from the University of Michigan. This wasn’t anything recreational or artistic, though, but instead a research and development project working with the glass capillary x-ray optics group at CHESS, the Cornell High Energy Synchrotron Source. For years the CHESS glass group has been pioneering a process of heating and tapering hollow glass tubes to create a sort-of “funnel” for x-ray light. Capillaries can make small, micron-sized x-ray beams that can be used to probe buried layers in paintings for art historians, uncover fingerprints of air pollution in the growth rings of trees, or scan tiny regions of miniature protein crystals to help solve puzzles in membrane biology.
Read more here... (html)
10/11/2007
_______________________________________________
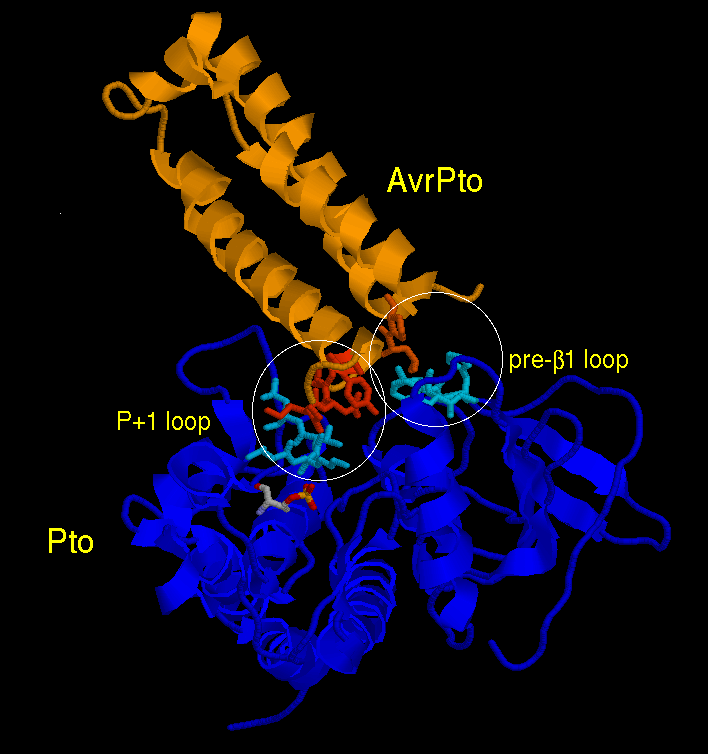
Structure of AvrPto/Pto Complex Helps Explain Plant Hypersensitivity
In the phenomenon called "hypersensitivity", plants seek to limit spread of a pathogen by destroying the affected region of plant tissue. The process involves recognition of a pathogen protein by a plant resistance protein. The first crystal structure of this type of recognition complex, between the bacterial protein AvrPto and the plant protein Pto, has now been determined by the Chai lab of the National Institute of Biological Sciences, Beijing, revealing important molecular details of the recognition process.
MAD data from a selenomet derivative were collected at the Beijing SRF. Structure solution was difficult, and MacCHESS staff members Quan Hao, Qingqiu Huang, and Qun Liu were called upon to assist with the processing. A 3.2 Å structure was finally obtained, with an R/Rfree of 27.1/30.3%. The interaction region between AvrPto and Pto is quite small (see figure); it involves one end of the mostly-helical AvrPto molecule and two separate loops on the surface of Pto.
Biochemical examination of Pto mutants, and structural comparisons with related proteins, enabled the authors to conclude:
Interaction with both of the Pto loops is required for
AvrPto to bind.
* Also, Pto must be in its active form, with a
phosphorylated threonine residue on the surface close to the interaction
region.
* Binding of AvrPto acts to inhibit, rather
than activate, Pto. This results in the activation of another
protein, Prf (which is normally repressed by Pto), leading finally to
the hypersensitivity response.
Read more here... (html)
8/28/2007
_______________________________________________

REU Undergraduates Innovate for Future Light Sources
It was certainly no easy vacation for 14 undergraduates who chose to spend their summer working on a wide variety of projects covering areas of particle physics, accelerator design and ways to improve x-ray optics for synchrotron research. They came to Cornell as part of the 2007 Research Experiences for Undergraduate (REU) program, funded by the National Science Foundation, through an interlocking grant between principal investigators Rich Galik, professor of Physics at Cornell, and Claude Pruneau, professor of Physics at Wayne State University. Like the 150 students over the decade before them, this year’s applicants found no time for a leisurely pace on the job.
Read more here... (html)
8/22/2007
_______________________________________________

Second CHESS Ph. D. Thesis Prize to Chemist Daniel Blasini
“Fuel cells as energy sources” and “organic light-emitting diodes” are keen interests of one of Cornell’s newly-minted physical chemists, Daniel Blasini. His graduate work at Cornell won the second annual Ph. D. Thesis Prize by the Cornell High Energy Synchrotron Source (CHESS). His graduate thesis, “Characterization of thin films and thin film phenomena in electrochemically active systems via x-ray methods,” documented five years of work focused on detailed surface structure and function of materials used in the high technology world of fuel cells, batteries and displays. During this time, Daniel developed methods and instruments to probe the physical chemistry of newly synthesized materials using powerful synchrotron x-ray beams.
More... (html)
(pdf)
Journal
of Materials Chemistry article: (pdf)
8/20/2007
_______________________________________________
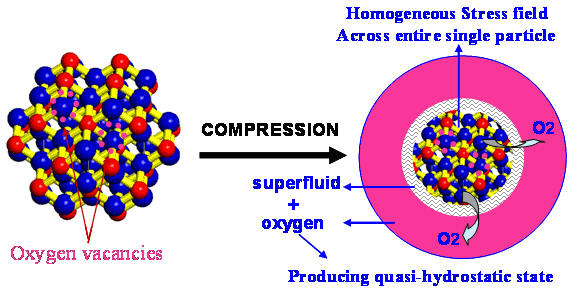
Smaller is More Bizarre in the Nanoworld of Ceria
Attention home builders: build your next house using 3 nanometer particles of cerium oxide (CeO2) and it will remain stable up to atmospheric pressures of 65 billion Pascals!
As we learn more and more about the small, nanoscale world around us, scientists and engineers are proposing and building new devices and components from nanoscale materials daily. Before we start building nano-houses and nano-robots, though, lots of challenges and hurdles stand in our way. Foremost among them is a common observation that many materials behave in unpredictable and bizarre ways when they get very small.
More... (html)
8/1/2007
_______________________________________________
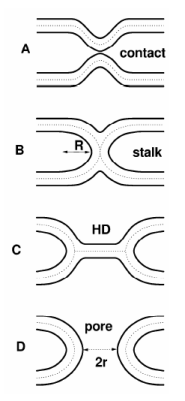 Well
known hypothetical fusion intermediates. Lipid
bilayer surfaces are indicated by solid lines.
Dotted lines in the hydrocarbon interior divide the bilayers into monolayers. A) Contact of the virus and target membranes B) Stalk that allows lipid mixing C) Hemifusion diaphragm (HD) and D) Pore.
Well
known hypothetical fusion intermediates. Lipid
bilayer surfaces are indicated by solid lines.
Dotted lines in the hydrocarbon interior divide the bilayers into monolayers. A) Contact of the virus and target membranes B) Stalk that allows lipid mixing C) Hemifusion diaphragm (HD) and D) Pore.
X-ray Study Reveals How HIV Infiltrates Cell Membranes
Contact: E. Fontes (ef11@cornell.edu)
Stephanie Tristram-Nagle and John F. Nagle (Carnegie Mellon) have used small-angle x-ray scattering at CHESS to uncover details about HIV infection. Quoting the press release from CMU - “Carnegie Mellon University scientists have made an important discovery that aids the understanding of why HIV enters immune cells with ease. The researchers found that after HIV docks onto a host cell, it dramatically lowers the energy required for a cell membrane to bend, making it easier for the virus to infect immune cells. The finding, in press in Biophysical Journal, will provide vital data to conduct future computer simulations of HIV dynamics to help further drug discovery and prevent deadly infections.”
"We found that HIV fusion peptide dramatically decreases the amount of energy needed to bend a cell-like membrane," said Stephanie Tristram-Nagle, associate research professor of biological physics at Carnegie Mellon. "This helps membranes to curve, a necessary step for HIV to fuse with an immune cell as it infects it."
The work was supported by the General Medicine Institute of the National Institutes of Health (GM 44976) and the CHESS is supported by the National Science Foundation (DMR-0225180)
Read the press release and report highlighted on
lightsources.org:
http://www.lightsources.org/cms/?pod=102255 or (pdf)
Published in Biophysical Journal, BioFAST: First Published May 25, 2007, doi:10.1529/biophysj.107.109181; Stephanie Tristram-Nagle and John F. Nagle; "HIV-1 Fusion Peptide Decreases Bending Energy, Promotes Curved Fusion Intermediates" (pdf)
7/26/2007
_______________________________________________

New X-ray Microscope Uncovers Mysteries in Famous Wyeth Painting
A new "confocal X-ray fluorescence (CXRF) microscope” has been developed at Cornell that lets art historians uncover minute details about buried features without risking damage to precious works of art. This CXRF microscope is the first of its kind at a powerful synchrotron source in the United States, and was built at the Cornell High Energy Synchrotron Source (CHESS) by an interdisciplinary team led by staff scientist Arthur Woll. Read more...
See the original article in the Cornell Chronicle : http://www.news.cornell.edu/stories/July07/
chessWyethAnalysis.html
See coverage also in the Cornell PawPrint issue July 19th (archived pdf here): http://www.pawprint.cornell.edu/docs/PP_July_19_web_07.pdf
Also on PhysOrg.com: (archived pdf):
http://www.physorg.com/news104161219.html
7/23/2007
_______________________________________________
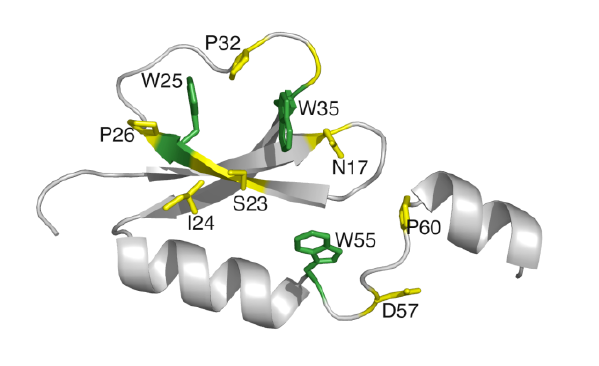
HWI Researchers Tackle Cystic Fibrosis and Tuberculosis
The structure of a novel protein in the bacterium that is the most persistent pathogen in cystic fibrosis (CF) patients has been solved. Hauptman-Woodward Institute scientists Dr. Andrew Gulick, Eric Drake and Dr. Manish Shah, in collaboration with researchers at SUNY Buffalo and the New York State Center of Excellence in Bioinformatics and Life Sciences have made this significant discovery, which was published in the current issue of the Journal of Biological Chemistry.
7/18/2007
_______________________________________________

Fast but Smooth - New Growth Method for Organic Films
The Materials Research Society (MRS) awarded a “best poster prize” to four collaborative groups at Cornell for their work “In-situ Real-Time Synchrotron Study of Small Molecule Organic Thin Film Growth from Hyperthermal Molecular Beams”, which reported the unexpected behavior of thin films of an organic semiconductor. The effort was headed by James Engstrom, faculty in the Cornell School of Chemical and Biomedical Engineering. This award was one of six bestowed on over 500 posters viewed by thousands of attendees at the San Francisco meeting held April 9-14, 2007.
7/9/2007
_______________________________________________
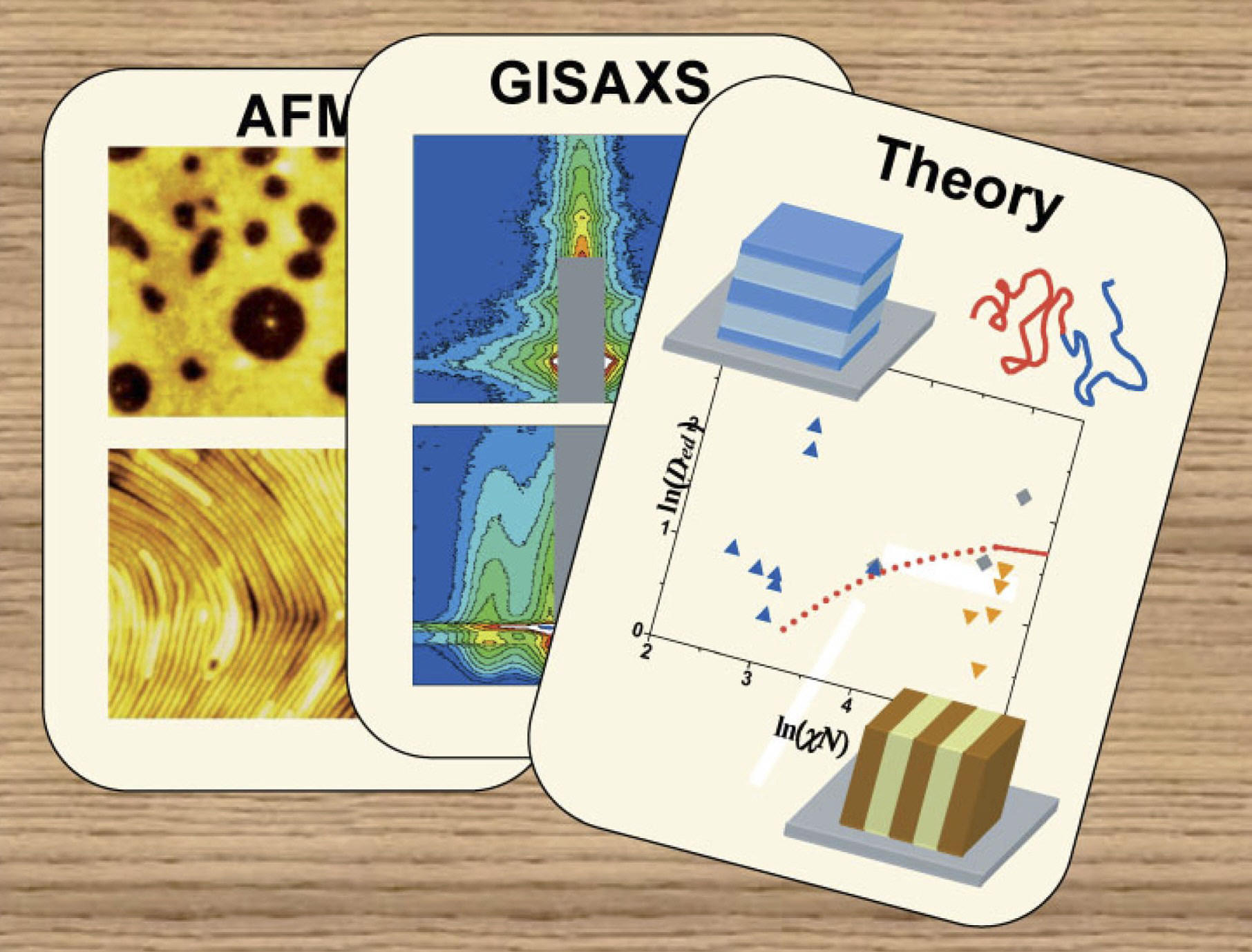
Diblock Copolymer Film Alignment Explained
For the May 2007 cover of journal Macromolecular Rapid Communication, CHESS staff scientist Detlef Smilgies designed artwork reflecting how two experimental techniques and one theoretical assault came together to unravel the ordering behavior seen in thin films composed of large molecular weight diblock copolymers.
More... (html)
Macromolecular
Rapid Communications (pdf)
Macromolecular
Rapid Communications Cover (pdf)
6/27/2007
_______________________________________________
New Method from CHESS to Record Thermal Distortions of X-ray Optics
Not a single biologist, chemists or physicist would use synchrotron radiation sources if not for high-quality optical elements that select, shape, and focus high-power, high-intensity x-ray beams onto their specimens. The challenge for the optics engineers, however, is providing the necessary dispersion and focusing all while dissipating the intense raw power in the full white beam. At the extremes, modern insertion devices driven by high energy particle beams produce x-ray broadband power densities equal to those produced by arc welders!
6/20/2007
_______________________________________________

Brian R. Crane
Protein Conformations Linked to Signal Propagation
On-line journal SpectroscopyNow highlights work by Cornell’s Crane group to understand the molecular affects of circadian rhythms. “Crane and his colleagues at Dartmouth Medical School, Hanover, New Hampshire, have studied the filamentous fungus, Neurospora crassa and revealed how circadian light sensors control production of carotenoids in this fungus… "We were interested in trying to understand behavior at the molecular level," said Crane. "This a great example of chemical biology, in that we can perturb the chemistry of a single molecule in a particular way and actually change the behavior of a complex organism."”
Lightsources.org has a science highlight at http://www.lightsources.org/
Crane's web site is at: http://www.chem.cornell.edu/faculty/index.asp?fac=23
6/5/2007
_______________________________________________
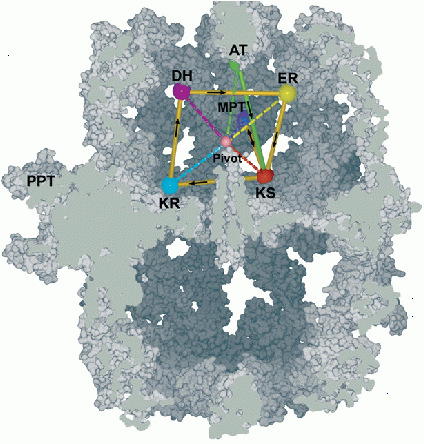
Fatty Acid Synthase Structure and Mechanism
Fatty acid synthase (FAS) type I from yeast is a 2.6 megaDalton complex which contains large internal reaction chambers, inside which a whole series of steps in the synthesis of fatty acids occurs. Fatty acids are major components of cell membranes and also serve as energy stores, and disruption of their synthesis causes severe metabolic disorders.
The Steitz group has now determined a 4 Å structure of yeast FAS I, using data from several synchrotron sources and the molecular replacement method (starting from a fungal FAS model). Due to 9-fold non-crystallographic symmetry and careful refinement, this rather low resolution structure has been highly informative about how this type of FAS works. In particular, it reveals the various positions occupied by the acyl carrier protein (ACP) domain as it moves the growing fatty acid chain between priming, elongation, and termination sites within the reaction chamber.
The figure shows a cutaway view of FAS, with the various active sites in one reaction chamber marked by colored dots. ACP is primed by loading with acetate at station AT, and then carries out multiple cycles around the ring KS, KR, DH, ER, and back to KS. During each cycle the motion of ACP is mainly a pivoting about the pink point in the center. When the fatty acid chain is long enough, it is transferred from ER to MPT for termination and release.
See full article (html)
5/31/2007
_______________________________________________
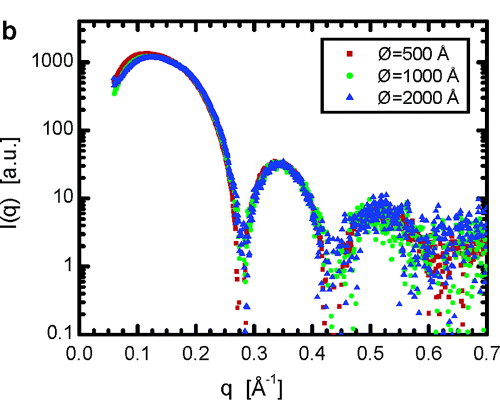
Nagle Group's Work Reveals Details of Lipid Bilayer Structure
John Nagle, with coworkers Stephanie Tristram-Nagle, Norbert Kuĉerka, and others, is continuing a decades-long study of the structure of phospholipid bilayers, which are simplified forms of biological membranes. This work is complementary to examination (by X-ray crystallography) of the structures of the proteins embedded in such membranes and is important in understanding how cells control their shape, permeability, and surface interactions.
An example of the group's recent work is the use of small-angle X-ray scattering (SAXS) data and sophisticated modeling to obtain a close look at the structure of fully hydrated fluid phase DPPC (dipalmitoylphosphatidylcholine) bilayers (Kuĉerka et al., Biophys. J. (2006) 90, L83-5). Since this is a fluid system, it is not possible to locate the positions of individual atoms; instead, overall properties, including the electron density profile across the bilayer and the area per phospholipid molecule, are determined. The paper reports experimental values for these properties, which will serve as important checks on future modeling efforts.
In another study (Kuĉerka et al., Langmuir (2007) 23, 1292-1299), SAXS, in combination with neutron scattering and dynamic light scattering, was used to examine the effect of curvature on bilayers, clearly a critical consideration for motile cells, which must change shape in order to move. Comparison of scattering from vesicles of different sizes (see figure) showed no effect of curvature on the thickness of bilayers composed of DOPC (dioleoyl-phosphatidylcholine), a neutral phospholipid. The work also clarified differences in the behavior of charged and neutral bilayers.
See full article (html)
3/30/2007
_______________________________________________

Dr. Darren Dale
Dr. Darren Dale Joins Ranks of CHESS Staff Scientists
Dr. Darren S. Dale was promoted to the rank of Staff Scientist at the Cornell High Energy Synchrotron Source (CHESS) on September 16th, 2006. Before that, Dale had been a post-doctoral associate at the laboratory following his Ph.D. thesis studies in the Materials Science and Engineering (MS&E) department at Cornell University.
See full article (html), (pdf)
3/05/2007
_______________________________________________

Showing lantern slides at a Gordon
Conference, 1957
(Photo: www.frontiersofscience.org historical archive)
Young Scientists Invited to X-ray Physics Gordon Conference
The next Gordon Research Conference (GRC) on X-ray Physics will be held August 5-10, 2007 at Colby-Sawyer College in New Hampshire. With an international cast of speakers, conference participants will hear about exciting X-ray-based science at 3rd generation light sources and scientific plans and early results from 4th generation sources.
2/26/2007
_______________________________________________

Grazing-incidence X-ray diffraction
pattern from an amorphous film
(see article, Figure 1A)
Amorphous Organic Films Show Medium-range Order that Bodes Well for Future Organic Devices
Cornell chemist Héctor Abruña tells readers of the Journal of Materials Chemistry how advances in organic light emitting diodes (OLEDs) depend upon synchrotron radiation studies similar to this done at CHESS. He starts his interview with:
“The performance of devices such as organic light emitting diodes (OLEDs) that depend on thin films are strongly correlated with the film's microstructure. While single crystal films generally exhibit the highest transport rates and quantum efficiencies, there are no cost effective methods to deposit such films over large areas. The use of polycrystalline films presents great challenges due to their high surface roughness and the fact that grain boundaries serve as efficient trapping and recombination sites resulting in dramatic degradation of performance. The use of amorphous films is most attractive because of their ease of deposition and because their hopping mode of transport is often sufficient for OLEDs and photovoltaic devices. Thus, an understanding of the local structure of nominally amorphous films could provide a great deal of insight as to how film morphology affects device performance. Our results provide a first characterization of a nominally amorphous molecular semiconductor thin film, and indicate that ordering actually occurs on an intermediate range, with crystalline domains in the range of a few nanometers. Such intermediate range order has been a hot topic in the community studying glassy materials in recent years.”
Full article: (html)
See Abruña's full comment archived - pdf
And the actual paper: (archived pdf):
http://www.rsc.org/delivery/_ArticleLinking/
ArticleLinking.cfm?JournalCode=JM&Year=2007
&ManuscriptID=b700505a&Iss=Advance_Article
2/23/2007
_______________________________________________

Surendra K. Saxena
Surendra K. Saxena wins 2007 Rudbeck Medal
Surendra K. Saxena, professor of Mechanical and Materials Engineering at Florida International University (FIU) in Miami, was awarded a Rudbeck Medal by Uppsala University in Sweden on January 26th, 2007. The award acknowledges his career of innovative research into high-pressure phenomena and high-pressure materials related to the Earth’s core.
"Olaus_Rudbeck." Wikipedia, the free encyclopedia. 16 Feb. 2007 http://www.reference.com/browse/wiki/Olaus_Rudbeck
Florida International University - http://www.eng.fiu.edu/cec/news/CEC_News_Surendra.htm
FIU archived:
(pdf)
2/19/2007
_______________________________________________

The picture shows the GaAs photocathode for the ERL
photoemission electron gun. The silver circle is the
active
area of the wafer that emits electrons when
exposed
to light to form the electron beam for the
accelerator.
The purple area around it is also GaAs,
but has been anodized
(coated with an oxide layer) to
prevent electron emission
from the edges off the wafer
due to scattered light.
[from: Bruce Dunham]
Design of Powerful New X-ray Source Moving Ahead
(from
article by Anne Ju, Cornell Chronicle Feb. 2, 2007)
Sol Gruner and Joel Brock provide an update on how New York State funds are helping the design effort underway for a first-of-its-kind X-ray source that will upgrade the existing Cornell facility, CHESS. The Cornell Chronicle published a short article with these highlights:
“Though the university hopes the National Science Foundation eventually will support the $300 million to $400 million synchrotron radiation facility at Cornell, called the Energy Recovery Linac (ERL), it is New York state funds that have proved instrumental in the early stages of the proposal's development.”
"The $12 million is for the kinds of things you need to put a solid proposal together," said Sol Gruner, professor of physics and director of X-ray science for the Cornell Laboratory for Accelerator-Based Science and Education (CLASSE), which oversees the ERL project. "So far, the allocation has paid for things the NSF would not have funded."
Joel Brock, professor of applied and engineering physics who is part of the design team for the ERL, said the device would "transform the research landscape at Cornell," and would be one of the university's largest capital undertakings in history.
Read the Cornell Chronicle article:
http://www.news.cornell.edu/stories/Feb07/erl.budget.aj.html
archived:
(pdf)
2/15/2007
_______________________________________________

Structure of the Zwit-1F beta-peptide bundle as
determined
by x-ray crystallography. The bundle
contains eight copies
of the beta-peptide Zwit-1F with
parallel and antiparallel helices
in like and unlike
colors, respectively.
[Image: Douglas S. Daniels]
Man-made Proteins Could Be More Useful than Real
Ones
(from HHMI press release, Feb. 6, 2007)
Researchers led by Alanna Schepartz, Yale University professor, HHMI investigator, and CHESS user, have constructed a protein out of amino acids not found in natural proteins, discovering that they can form a complex, stable structure that closely resembles a natural protein. Their findings could help scientists design drugs that look and act like real proteins but won't be degraded by enzymes or targeted by the immune system, as natural proteins are.
"The fundamental insight from this study is that β-peptides can assemble into structures that generally resemble natural proteins in shape and stability," Schepartz said. She added that their findings about the structure of the molecule that she and her colleagues synthesized will help scientists construct more elaborate β-peptide assemblies and ones that possess true biologic function.
Such β-peptides could also be designed as pharmaceuticals that would be more effective than natural protein drugs, because the enzymes that degrade natural proteins would not affect them.
See the HHMI article:
http://www.hhmi.org/news/schepartz20070206.html,
archived: (pdf)
Or article in ACS Chemical & Engineering News:
http://pubs.acs.org/cen/news/85/i06/8506notw1.html
archived: (pdf)
Or lightsources.org highlight:
http://www.lightsources.org/cms/?pid=1001921
archived: (pdf)
2/7/2007
_______________________________________________
"Watching Atoms Move" - Brock views future ultrafast X-ray science
Contact: E. Fontes (ef11@cornell.edu)
Joel Brock, professor of Applied and Engineering Physics at Cornell, published a February 2nd commentary in the journal Science about how future X-ray sources will address the difficult challenge of visualizing motions of molecules and atoms. He introduces and highlights a research report, published in the same issue, from an international collaboration of researchers who used a prototype X-ray machine at Stanford to observe the oscillation of excited atoms in a bismuth crystal. The prototype machine, called Sub-Picosecond Photon Source (SPPS), is now being replaced by a full-scale X-ray free-electron laser (XFEL), the Linac Coherent Light Source (LCLS). Brock discusses how free-electron lasers, and the next generation Energy Recovery Linac (ERL) source (now being designed as an upgrade to CHESS), will be magnificent machines - far brighter and able to produce much faster, sub-picosecond, X-ray flashes than today’s storage rings. He speculates that scientists will excitedly progress from “still photos” to “movies” of dynamic atoms and molecules.
See the Cornell Chronicle article:
http://www.news.cornell.edu/stories/Feb07/brock.atoms.aj.html,
archived: (pdf)
See the Science article (may require subscription):
http://www.sciencemag.org/cgi/content/full/315/5812/609
archived: (pdf)
2/6/2007
_______________________________________________

Nominees Sought for Annual CHESS Ph.D. Thesis Prize
CHESS is now accepting nominees for a 2nd Annual CHESS Thesis Prize of $1,000. The award will go to a student receiving a PhD or Masters degree during the year March 2006 to March 2007. The thesis work must be based in part on X-ray research done using the Cornell High Energy Synchrotron Source.
For more information: (html), (pdf)
2/6/2007
_______________________________________________

John Ferguson
G-line Students Show off X-ray Projects
G-line scientist Arthur Woll organized a night of science, food and discussion for Cornell graduate students who do X-ray work at CHESS, the Cornell High Energy Synchrotron Source. G-line is a set of three high-intensity X-ray stations that were built by graduate students a few years ago, and continue to be maintained by students to serve the research needs of the Cornell Community. This symposium is held twice a year, giving the CHESS community an update on the accomplishments of the local faculty groups and student projects. This sixth meeting in the series was held Tuesday evening, January 23rd, 2007 in the Commons area of the Wilson Laboratory. Pizza, soft drinks, and coffee helped sustain the agenda!
1/31/2007
_______________________________________________
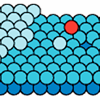
DNA nucleotide
Virus Surfaces and Gene Therapy
The Agbandje-McKenna group investigates the structures of adeno-associated virus (AAV), which is a candidate for DNA carrier in gene therapy. Different types of AAV vary in their affinity for different types of cells, and the research seeks to correlate viral surface structures with cellular specificity.
The recent determination of the structure of AAV4, done by molecular replacement, using data from CHESS F1 station, and its comparison with that of AAV2, showed that the core structure of the viral capsid remains the same among different AAVs, while surface features differ substantially. Knowledge of how surface structures can be changed without destroying the capsid will guide future genetic engineering work.
The figure shows a DNA nucleotide, bound in a pocket on the inside of AAV4. The presence of this ligand raises questions about the interaction between AAV (and related viruses) and DNA.
See article (html)
Journal of Virology Vol. 80, No.
23, for
December 2006
or read PDF archive article here.
1/24/2007
_______________________________________________
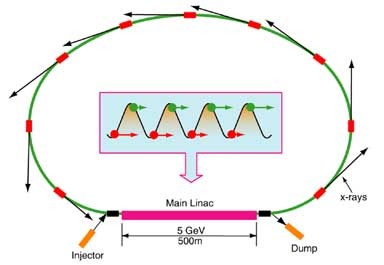
Schematic of an ERL light source
Status of Energy Recovery Linac Sources
The Synchrotron Radiation News published an article on the status of Energy Recovery Linac efforts at Cornell and worldwide. Authors Bilderback, Sinclair and Gruner discuss how linac-based sources, such at Energy Recovery Linacs (ERLs) and X-ray free electron lasers (XFELs) are at an early stage of development but provide a clear path towards dramatically improving X-ray beam qualities. These new sources will enable new techniques that use ultra-fast pulses, coherence, and extreme brightness - and should spur exciting new scientific discoveries.
See Synchrotron Radiation News Vol. 19, No. 6, for
November/December 2006
or read PDF archive article here.
1/5/2007
_______________________________________________
